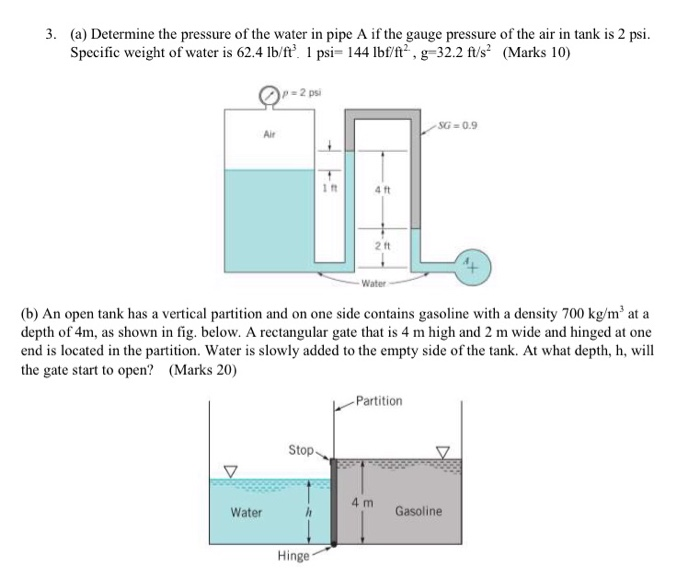  
The shearing stress on a plane parallel to the walls and passing through the centerline? at 2. What is the shearing stress at the bottom wall? 2. Determine the values of the Reynolds number using (a) SI and (BG) unitsġ7 Figure E1.5 (p. For gases, For liquids, Kinematic viscosity units are / ft2 m2 /sĪ Newtonian fluid having a viscosity of 0.38 N.s/m2 and a specific gravity of 0.91 flows through a 25 mm diameter pipe with a velocity of 2.6 m/s. 17) Dynamic (absolute) viscosity of some common fluids as a function of temperature. Units of Viscosity – lb.s/ft2 N.s/m2ġ5 Figure 1.6 (p. 16) Variation of shearing stress with rate of shearing strain for several types of fluids, including common non-Newtonian fluids. μ= absolute or dynamic viscosityġ4 Figure 1.5 (p. Linear variation of shearing stress with rate of shearing strain for common fluids. Shear stressτ occurs at the plate-material interface at equilibrium, P=τA velocity gradient, du / dy =U/b Fluid sticks to the wall=no-slip condition as Rate of shearing strain 14) Behavior of a fluid placed between two parallel plates. (b) Forces acting on upper plate.ġ2 Figure 1.3 (p. 13) (a) Deformation of material placed between two parallel plates. A fluid with low viscosity flows easily because its molecular makeup results in very little friction when it is in motion.ġ1 Fluid motion can cause shearing stressesįigure 1.2 (p.A fluid with large viscosity resists motion because its molecular makeup gives it a lot of internal friction. It describes the internal friction of a moving fluid. When the tank is filled with air at a gage pressure of 50 psi, determine the density of the air and the weight of air in the tank.Ĩ Example 3 Determine the density, specific gravity and mass of the air in a room whose dimensions are 4 m x 5 m x 6 m at 100 kPa and 25 Cĩ Viscosity Viscosity is a measure of a fluid's resistance to flow. R LV5 Ts are expressed in Kelvin or Rankine Lisa Vink, ħ Example 2 A compressed air tank has a volume of 0.84 ft 3. R is different for each gas and is determined from R=Ru/M where Ru is the universal gas constant, Ru=8.314 kJ/kmol.K=1.986 Btu/lbmol. Gases are highly compressible, gas density changes with pressure and temperature as, where p is the absolute pressure, ρ the density, T is absolute temperature, and R is gas constant Pressure units: lb/ft2 (psf) lb/in2 (psi) N/m2 (Pa) Standard sea-level atmospheric pressure – 14.7 psi kPa Gage pressure + atmospheric pressure = absolute pressureĥ Ts are expressed in Kelvin or Rankine Lisa Vink,.10) Density of water as a function of temperature.Ĥ Ideal/Perfect Gas Law /Equation of State Determine its density and specific weight at this temperature in both BG and SI units,ģ Figure 1.1 (p. Specific Gravity = density of the fluid/density of waterĢ Example 1 The specific gravity of mercury at 80 0C isġ3.4.Specific weight= weight/volume (lb/ft3 N/m3).Specific volume (volume per unit mass),.1 Density, ρ= mass/unitvolume –Slugs/ft3 kg/m3
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
